|
This table also includes data for these functions for the transfer of ions into aqueous urea, which, though urea is not a solvenf it behaves in aqueous solutions as if it were a liquid amide. Data are available in this reference also for many other aqueous solvents (n- and i-PrOH, f-BuOH, glycerol, tetrahydrofuran, 1,4-dioxane, acetone, A,A-dimethylacetamide, and sulfolane) for at least a part (the water-rich part) of the composition range, as well as for some other ions that were not measured atX3=0.5 in the solvents shown in Table 6.2 or that could not be traced to the TATB or TPTB assumptions. The ionic data were selected from data on electrolytes hsted in these references on the basis of their conformation to the TATB or TPTB assumptions (Section 4.3.1). Further values for the solvents shown there are available at 0.1 mole fraction steps over the entire composition range. Table 6.2 shows values of the standard molar enthalpy and entropy for the transfer of ions from water into equimolar aqueous mixtures with cosolvents at 25☌, A, /f or A j5 "(I, W 0.5W-i-0.5S), taken from the compilation by Hefter et al. 
The entropy contribution, TAs ° (kJ/C-mole), to the free enthalpy of combustion at standard conditions, as a function of the degree of reduction, y, of the compounds considered, for acids (S), carbohydrates (A), alkanes (O, ethene and ethyne (O), alcohols (9), acetone itM). The differential thermodynamic parameters of activation, with acetone taken as the standard reactant, are given in Table 14. A classic example of a reaction in which relative rates are largely determined by TAS terms is semicarbazone formation in phosphate buffers (Price and Hammett, 1941). The role of entropy in determining the order of reactivity within a reaction series has been demonstrated for several other reaction systems. Standard Free Energy, Enthalpy, and Entropy of Transfer of n-Bu4NBr from Water to Water-Acetone Mixtures at 298.15°K. 3) (c) the standard free energy of vaporization (d) the boiling point of 2-propanone. Calculate (a) the standard enthalpy of vaporization (b) the standard entropy of vaporization (use Eq. The vapor pressure of 2-propanone (CH COCH, more commonly known as acetone) is 67 Torr at 0.0☌ and 222 Torr at 25.0☌. 5, its standard entropy of vaporization at its boiling point is. To calculate the standard entropy of vaporization of acetone at its boiling point of 329.4 K (corresponding to 56.2☌), we note from Table 6.2 that its standard enthalpy of vaporization at its boiling point is 29.1 kjmol-1. 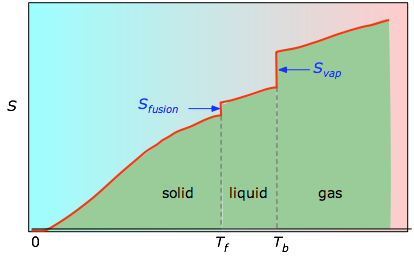
The figure was constructed as follows first the standard enthalpy of transfer AH°t, obtained by Ahluwalia and co-workers (12) from pure water to Z2 = 0.30, was used in order to get the standard entropy of transfer function from the relation. vaporization is different in form from the method outlined by Giauque and Kemp.2 Their approach leads to two equations, the one expressing the two unknowns in terms of the entropy of dissoci ation of the dimer, the other relating them to the entropy of the mixture and the entropy of mixing. 
Figure 4 and Table IV present the results obtained. We shall discuss now the variation of the three main thermodynamic functions with solvent composition for the case of n-Bu4NBr- water-acetone system and shall extend this discussion to the n-Bu4NBr-water-THF system. g of acetone, CH3COCH3, condenses at its normal boiling point. (b) What is the entropy change of the surroundings when 10. The standard entropy of vaporization of acetone is approximately 85 J-K l-mol at its boiling point, (a) Estimate the standard enthalpy of vaporization of acetone at its normal boiling point of 56.2☌. Note that the enthalpies of fusion and vaporization change with temperature.What is the standard entropy of vaporization of acetone at its normal boiling point of 56.2☌. Solids like ice which have strong intermolecular forces have much higher values than those like CH 4 with weak ones.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
